Compiled by: Slawomir Chorek
Bonding technique
A short course in basic concepts.
Table of contents:
Adhesion
Adhesion is defined as the forces of adhesion between the surfaces of different materials. The physical forces of attraction or adsorption, known as Van der Waals forces (Van der Waals forces are long-range attractive interatomic or intermolecular interactions), are of primary importance in adhesion.
The interaction range of these intermolecular forces is much smaller than the mechanical roughness of the treated surfaces. Therefore, the adhesive must penetrate the roughness and completely wet the surfaces.
The ability of a liquid adhesive to wet the surface depends on the surface energy of the materials. The adhesive will only sufficiently wet the surface of a material if its surface tension is at least equal to or less than the surface energy of the material. Examples of the surface free energies of different materials at room temperature:
- PTFE (Teflon): 18 mN/m
- PVC : 40 mN/m
- Polyamide 6/6: 46 mN/m
- Iron: 2030 mN/m
- Tungsten: 6800 mN/m
for comparison:
- anaerobic adhesives: 30-47 mN/m
The table shows that metals are easy to bond, whereas plastics are often critical. Appropriate pre-treatment can have a positive effect on the surface energy of the parts to be bonded.
Adhesive failure occurs when the adhesive forces are less than both the cohesion forces and the external load.
Cohesion
The cohesion of an adhesive bond is defined by its mechanical strength, which is a manifestation of the mutual attraction of the adhesive particles. Destruction of the bond can occur as a result of
- Detachment of the adhesive layer from the substrate – Adhesive failure
- Destruction of the adhesive film – cohesive destruction
- Destruction of the bonded material – structural failure.
Cohesive failure occurs when the intermolecular bonding forces of the adhesive give way to the cohesion force and the external load force.
According to the rule that a chain is only as strong as its weakest link, the adhesive and cohesion forces should be of similar magnitude.
Surface preparation for bonding
The stronger the bond, the more thoroughly the surfaces in the bond area have been cleaned.
Bond strength can be significantly increased by
- Removal of foreign surface layers by degreasing or mechanical treatment.
- Formation of new active surface layers by chemical treatment.
Degrease mating surfaces
The basis of a reliable joint is the complete removal of oil, grease, dust and other contaminants from the surfaces to be bonded, as these reduce the holding forces at the joint. All solvents that evaporate without trace from washed surfaces are suitable for this purpose.
Mechanical treatment
Metal surfaces often have an oxide layer that cannot be removed by degreasing alone.
It is then recommended that the surface be mechanically treated by sanding, grinding or roughening.
When sanding with sandpaper, make sure that the grit size is appropriate (e.g. 300 to 600 for aluminium, 100 for steel).
After any of these methods, the parts to be bonded should be thoroughly degreased. When bonding rubber parts it is recommended to mechanically remove any pressed or vulcanised layers.
Sandblasting plastics with hardened iron or electro-corundum powders has been found to be effective.
Rubber surfaces should be stripped of anti-adhesive agents using a solvent or sandpaper.
Chemical treatment
Some materials that are difficult to bond (especially plastics) require chemical treatment. The most common treatment is surface etching. The composition of etching baths is recommended by adhesive manufacturers in their instructions.
Examples of correct bonding
The properties and capabilities of adhesives should be considered at the design stage. For optimum results, only shear and/or compression forces should be applied to the bond.
Peel forces have an extremely negative effect on the bond, but can be avoided by modifying the design. The surfaces should be as large as possible in order to transmit high forces.
During assembly, care must be taken to ensure that the applied adhesive is not pushed off the parts. Chamfer the parts to be joined (chamfer angle 15-35°). It is also necessary to check the mutual positioning of the parts, as correcting them during the curing process will destroy the polymerisation chains formed, affecting the final strength of the bond.
When fitting parts into a blind hole, the compressed air in the hole can push the adhesive off the part being fitted. This can be avoided by using a vent hole or by dispensing the adhesive directly into the blind hole. When the mating part is pressed in, the adhesive is pushed up and fills the gap well.
When bonding parts with very different coefficients of thermal expansion, large tensile forces can be generated at the joint when temperature differences occur. The adhesive can transmit such forces in the shaft-hub joint if the inner part has a higher coefficient of expansion. Otherwise an interference fit must be used to ensure that the hub does not slip off the shaft.
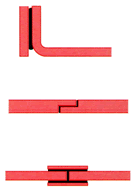

Direction of forces on bonded joints
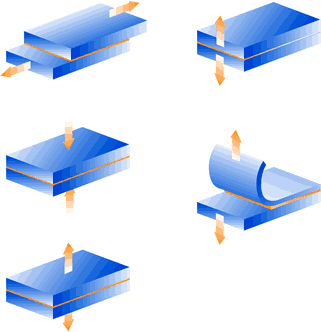
The figures on the left illustrate the correct distribution in a bonded joint. Structures with forces acting in the directions shown in the figures on the right (eccentric pull and peel) should be avoided.
Application methods for 2-part adhesives
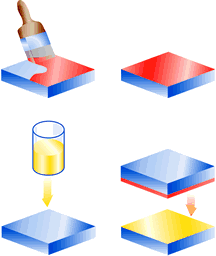
Bond with a hardener in the form of a ‘varnish’. One of the surfaces to be bonded can even be prepared well in advance of the bonding process by applying a layer of varnish hardener beforehand.
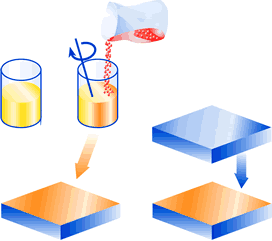
Hardener in paste or powder form is added to the adhesive immediately before bonding. The open time determines the time before the parts must be assembled. Some adhesives require very thorough (even) mixing of both components.
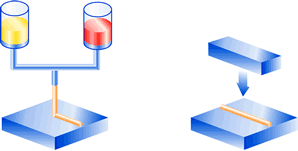
Dosing by means of a dispenser with automatic mixing of the two adhesive components.
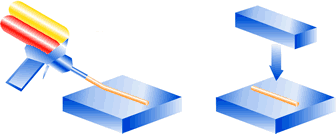
Glue in two chamber cartridges. Dispensing by manual or pneumatic gun. Cartridges provide a ‘ready made’, simple and cost effective dispensing system, replacing expensive dispensing equipment.
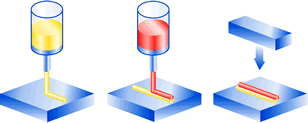
Bead-on-bead (drop-on-drop) adhesive application.
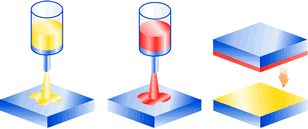
The classic A/B system. The adhesive components are applied separately to each surface and then joined together. The curing reaction only occurs when the two surfaces come into contact.
Contact Us
Please complete the form below and you will be contacted shortly.
For immediate technical assistance, please contact me on the following phone number:
601536440

